Guidelines for the development, implementation and use of data-driven precision medicine for all.
Precision medicine stems from progress in bio-medical sciences. It is expected to deliver numerous benefits to individuals by significantly improving the state of health of each, and thus of the entire population. However, its development presents some critical challenges, which require a clear strategy.
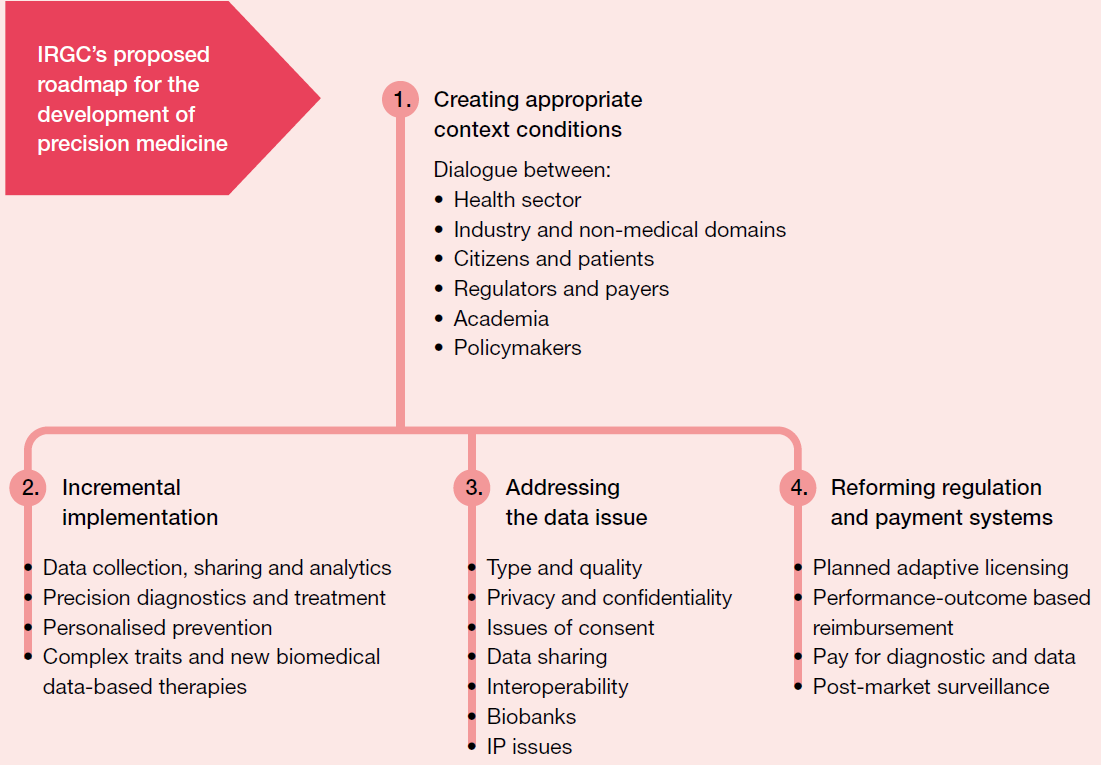
Following a one-day invitation-only expert workshop at Campus Biotech in Geneva on the Governance of Impacts of Precision Medicine, IRGC decided to produce a roadmap for precision medicine.The purpose of the overarching and comprehensive roadmap proposed by IRGC is to recommend a structured process that organisations can follow in their transition towards the development of precision medicine. The process is adaptable, and each organisation should further develop it to align with its own needs, culture and objectives.
The roadmap recommends a comprehensive approach to the development of precision medicine:
- It begins with the creation of a space for dialogue, to create favourable context conditions by engaging the many stakeholders involved.
- It considers a step-by-step implementation of precision medicine, depending on such issues as scientific development, cost and uncertainties involved.
- It recommends paying careful attention to the issue of data.
- It emphasises that regulation and payment systems will have to be adapted.
Conscious of differences in cultures, legal frameworks, social contexts, economic aspects or health priorities, this roadmap does not intend to prescribe certain specific policy and regulatory choices.
The recommendations in this roadmap primarily address an audience in government and other public institutions, which may use the roadmap as a set of generic guidelines for developing their own strategy for precision medicine. Other actors in the public and private sectors, such as patients associations, healthcare payers, civil society representatives, or professional and scientific associations may also find this roadmap helpful.

Download
- Roadmap for Precision Medicine (Policy Brief, 2017)
